This document provides information of cholesterol, a non-mysterious reagent that has been used in liposomes, LNPs and even adjuvants (AS01B). Its nature, structure and role are familiar to us. Let’s have a better understanding.
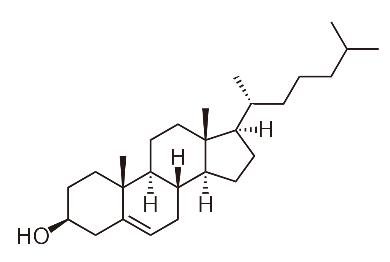

Fig. 1. Cholesterol
In 1769, French physician François Poulletier de la Salle first identified solid cholesterol in gallstones.
In 1815, French chemist Michel Eugène Chevreul named the compound “Cholesterol”.
(Its English name “cholesterol” is the Greek word “chole” (bile) plus “stereos” (solid), ending with “ol” (because of the hydroxyl group in its chemical structure).
Appearance: white crystalline powder | CAS: 57 88 5 |
Chemical formula: C27H46O | Density: 1.052 g/cm3 |
Molar mass: 386.65 g/mol | Boiling Point: 360°C (633 K) |
Melting point: 148-150°C (271 K) | Flash Point: 209.3 ± 12.4 °C |
Solubility: sparingly soluble in water (0.095 mg/L at 30°C); soluble in acetone, benzene, chloroform, ethanol, diethyl ether, hexane, isopropyl myristate, methanol. | |
Cholesterol accounts for approximately 30% of any animal membrane. Cholesterol constructs and maintains cell membranes and regulates membrane fluidity within physiological temperature ranges.
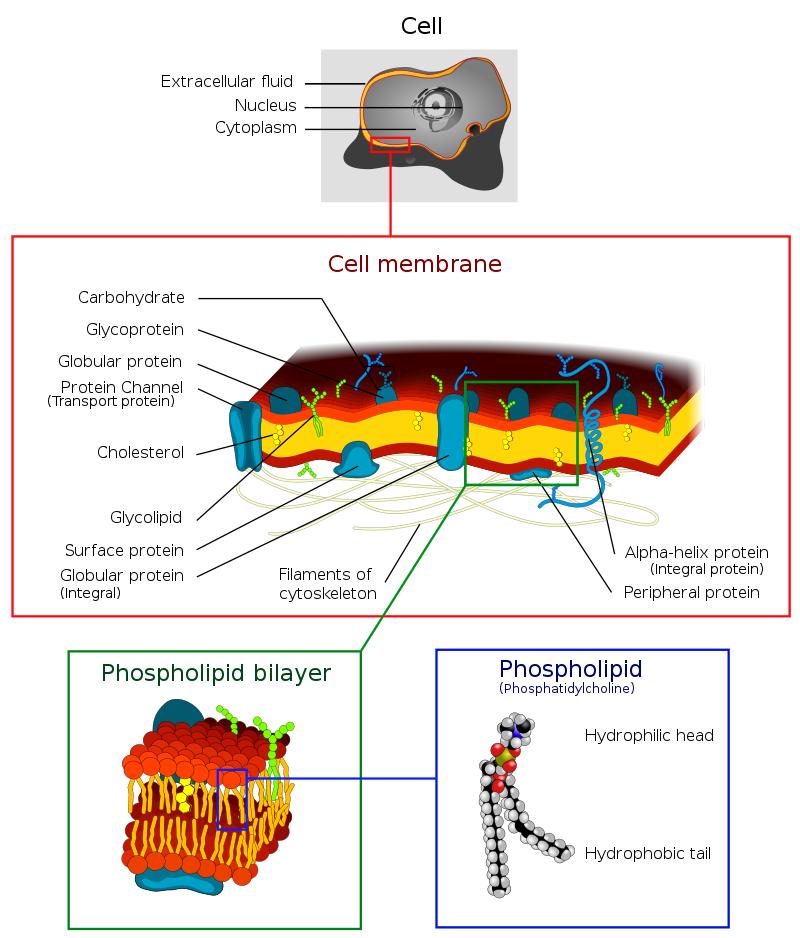
·The hydroxyl groups of each cholesterol molecule can interact with water molecules around the membrane, as do polar heads of membrane phospholipids and sphingolipids. The hydrocarbon chains of steroids and other lipids are embedded in the cell membrane together with non-polar fatty acid chains. By interacting with phospholipid fatty acid chains, cholesterol increases membrane packing, which alters membrane fluidity and maintains membrane integrity. Thus, unlike plants and most bacteria, cells of animals do not have cell walls. While the cell membrane remains stable and durable, it is not rigid, which allows animal cells to change shape and move.
·The structure of the cholesterol tetraloop contributes to the fluidity of the cell membrane. The molecule in a trans conformation allows all parts except the side chain of cholesterol to be rigid and planar. In response to this structure, cholesterol also decreases the permeability of the plasma membrane to neutral solutes, hydrogen ions, and sodium ions.
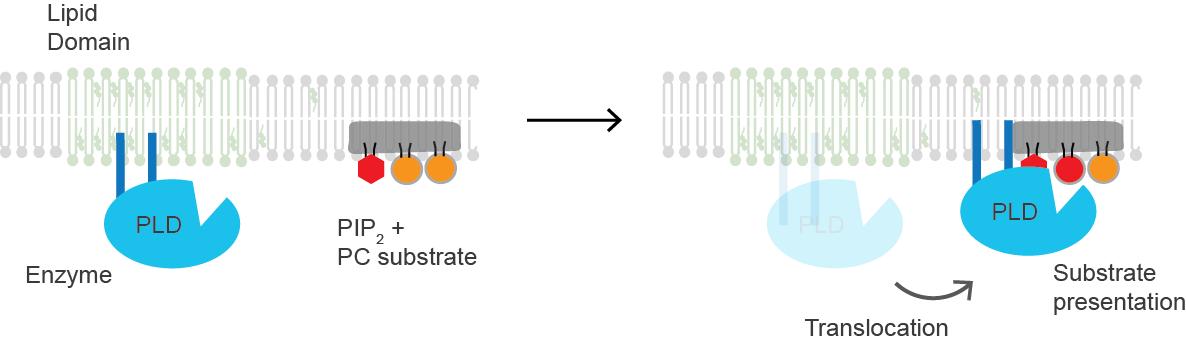
Cholesterol regulates the biological process of substrate presentation as well as enzymes activated by substrate presentation. Phospholipase D2 (PLD2) is an enzyme typically activated by substrate presentation. The enzyme is palmitoylated (covalently linked by palmitic acid and cysteine), causing it to be transported to cholesterol-dependent lipid domains called “lipid rafts”. The substrate of PLD2 is phosphatidylcholine (PC), which is unsaturated and is present in low abundance in lipid rafts. PC localizes to disordered regions of the cell along with the polyunsaturated lipid phosphatidylinositol 4,5-bisphosphate (PIP2), a substrate for many important signaling proteins. PLD2 has a PIP2-binding domain. When PIP2 concentration increases in the membrane, PLD2 leaves the cholesterol-dependent domain and binds to PIP2, and then binds to its substrate phosphatidylcholine in the PIP2-binding domain to catalyze the binding.
Cholesterol is also involved in the cellular signaling process, contributing to lipid raft formation in the plasma membrane and thus bringing receptor proteins into proximity with high concentrations of second messenger molecules. In multiple layers, cholesterol and phospholipids are electrical insulators that facilitate the speed of electrical impulse transmission along neural tissue. For many neuronal fibers, myelin is rich in cholesterol, providing insulation for more efficient impulse conduction. Demyelination is considered to be part of the basis of multiple sclerosis.
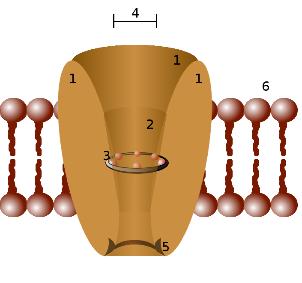
Cholesterol binds to and influences the gating of many ion channels, such as nicotinic acetylcholine receptors, GABAA receptors, and inwardly rectifying potassium channels. Cholesterol also activates estrogen-related receptor alpha (ERRα) and may be an endogenous ligand for this receptor. Inhibition of ERRα signaling by reducing cholesterol production has been identified as a key mediator of the effects of statins and bisphosphonates on bone, muscle, and macrophages. Based on these findings, it has been suggested that ERRα is de-orphaned and classified as a cholesterol receptor.
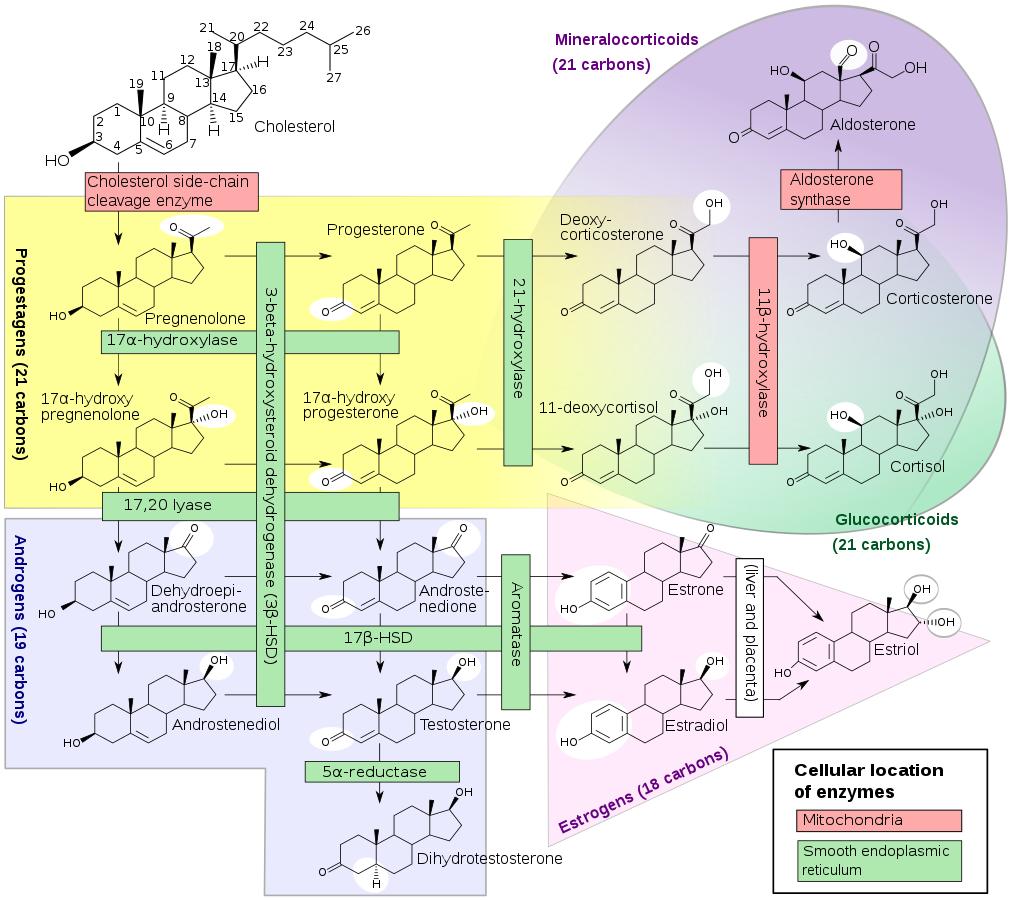
Intracellularly, cholesterol is a precursor molecule for several biochemical pathways. For example, it is a precursor molecule in calcium metabolism that synthesizes vitamin D and all steroid hormones such as adrenal hormones cortisol and aldosterone, as well as progesterone, estrogen, testosterone and their derivatives.
Cholesterol circulates in the body. Cholesterol is excreted into the bile by the liver, then stored in the gallbladder, and then excreted into the digestive tract in a non-esterified form (via bile). Typically, approximately 50% of the excreted cholesterol is reabsorbed back into the blood by the small intestine.
“Position” | “Responsibilities” |
Emulsifier | Cholesterol may be used as an emulsifier when making oil-in-water emulsions. |
Vaccine adjuvant | A liposome prepared from cholesterol is used as the adjuvant in AS01B to reduce hemolysis of QS-21. |
Ointment base | As a hydrophilic and absorbent ointment base, it may increase the emulsification and penetration of medicinal oils in pharmaceuticals. With emollient properties, it commonly used in formulations at concentrations of 0.3% to 0.5% (W/W) to increase the water absorption capacity of ointments. |
Membrane material for preparing liposomes | Cholesterol can be used as a membrane material with other phospholipid excipients to prepare liposomes. |
Lipid nanoparticles | Cholesterol can be combined with cationic lipids, structural phospholipids and PEGylated lipids to prepare LNP for the delivery of RNA drugs/vaccines and effectively improve the fluidity and stability of lipid membranes. |
Not finished | To be continued |
References:
[1]. Wikipedia, the free encyclopedia
[2]. PubChem (nih.gov)
[3]. Yeagle PL (October 1991). “Modulation of membrane function by cholesterol”. Biochimie. 73 (10): 1303–10. doi:10.1016/0300-9084(91)90093-G. PMID 1664240.
[4]. Incardona JP, Eaton S (April 2000). “Cholesterol in signal transduction”. Current Opinion in Cell Biology. 12 (2): 193–203. doi:10.1016/S0955-0674(99)00076-9. PMID 10712926.
[5]. Levitan I, Singh DK, Rosenhouse-Dantsker A (2014). “Cholesterol binding to ion channels”. Frontiers in Physiology. 5: 65. doi:10.3389/fphys.2014.00065. PMC 3935357. PMID 24616704.
[6]. Cohn JS, Kamili A, Wat E, Chung RW, Tandy S (February 2010). “Dietary phospholipids and intestinal cholesterol absorption”. Nutrients. 2 (2): 116–27. doi:10.3390/nu2020116. PMC 3257636. PMID 22254012.
AVT specializes in providing high-quality RNA-delivery excipients and LNP and Liposome excipients are timely and readily available.
AVT will always work with you on the long road to RNA delivery.
Now AVT has introduced plant-derived cholesterol. Contact us for more information.