D(+)-trehalose dihydrate is a stable non-reducing disaccharide. It can form a protective film on the surface of cells under various environmental stresses such as extreme heat, cold, drought, effectively to effectively protect the structure of biological macromolecules from damage. It has excellent non-specific protective function for biological cells, and can improve the resistance of biological cells to harsh conditions.
In recent years, D(+)-trehalose dihydrate has been widely used in biological products including antibody drugs. It is used as a protective agent for about 30% of antibody drugs on the market (including ADCs), such as Adcetris, Padcev, Imjudo, Kimmtrak, etc.
D(+)-trehalose dihydrate also plays an increasingly important role in cell cryopreservation, assisted reproduction and other fields. Here, AVT's product team interprets the following related literature to help you understand the relevant research progress.
D(+)-Trehalose Dihydrate for Cryopreservation of OPCs
Demyelinating diseases are neurological disorders characterized by loss of the myelin sheath, such as spinal cord injury (SCI), multiple sclerosis (MS), etc.
OPCs can differentiate into mature oligodendrocytes in vivo, which are important myelinating cells of the central nervous system and encapsulate neuronal axons to constitute myelin structures. Therefore, transplantation of OPCs is considered to be an highly potential therapy for demyelinating diseases.
Cryopreservation of OPCs is a critical step in successful transplantation of human OPCs.
Before being used clinically, human OPCs were cryopreserved with the cryoprotectant of 100 mL/L DMSO + 900 mL/L FBS (the method proposed in 1984), with a recovery rate of 55% to 70%. The method was not optimized for a long time thereafter.
Given that DMSO is cytotoxic and FBS is immunogenic, the above cryopreservation method is not suitable for clinical use.
In order to explore a more economical, effective, safe human OPC cryopreservation method that can be applied on a large scale, Liu Chang from Southern Medical University and other researchers optimized the above method using OPCs induced by human fetal brain neural stem cells (hNSCs).
① Comparing the effects of different methods on the recovery rate of OPCs, it was found that the survival rate of cells collected by digestion was significantly higher than that by mechanical pipetting.
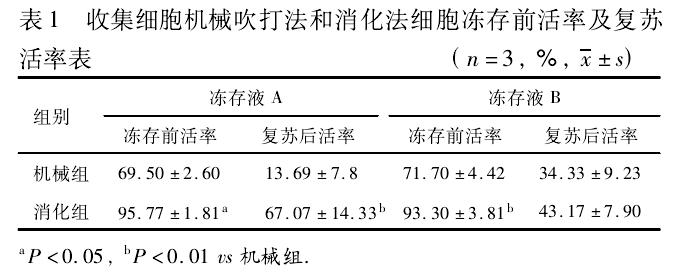
表1收集细胞机械吹打法和消化法细胞冻存前活率及复苏活率表
Table 1: Cell Viability and Recovery Rates Before and After Cryopreservation Using Mechanical Disruption and Digestion Methods
组别、机械组、消化组 Group Mechanical Group Digestion Group
冻存液A、冻存液B Cryoprotectant A Cryoprotectant B
冻存前活率、复苏后活率 Viability Before Cryopreservation Viability After Recovery
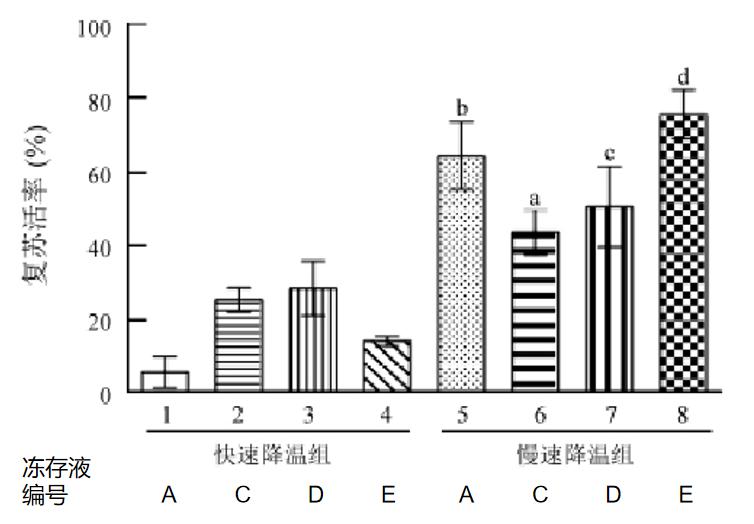
② Comparing the recovery rate of OPCs cryopreserved with four different cryopreservation media by slow cooling in the freezing box and fast cooling in the vapor phase liquid nitrogen tank.
No. | Cryopreservation medium composition |
A | Cryopreservation medium containing 70 mL/L DMSO + 930 mL/L FBS |
B | hPSC serum-free cryopreservation medium |
C | OPS complete medium containing 70 mL/L DMSO + 300 mL/L FBS |
D | OPS complete medium containing 70 mL/L DMSO + 100 mL/L FBS + 300 mL/L HES |
E | OPS complete medium containing 70 mL/L DMSO + 300 mL/L FBS + 0.2 mol/mL D(+)-trehalose dihydrate |
As shown below, there was a statistically significant difference in the recovery rates for the two cooling rates. The recovery rates for the four cryoprotectants were all less than 30% by rapid cooling. The recovery rate for cryopreservation medium E supplemented with D(+)-trehalose dihydrate was the highest by slow cooling in the freezing box, which was (75.73 ± 6.66)%.
③ Comparing the effect of the duration of cryopreservation on the recovery rate of OPCs
The cells cryopreserved in cryopreservation medium E with the highest recovery rate were selected for subsequent experiments, and the recovery rate was compared between short-term (<2 months) cryopreservation and long-term (6 months) cryopreservation, showing no significant difference in the recovery rate between the two groups.
④ Comparing the cell proliferation activity
Cryopreservation medium E with the highest recovery rate was selected for cell proliferation comparison. The mean proliferation multiple was compared between the cryopreservation medium E group and the normal culture control group after 3 times of serial passage at the same time, and there was no significant difference between the two groups. After 7 days of culture, there was no significant difference in the proliferation rate between the normal culture group and the post-thaw culture group. The results indicated that the cell proliferation activity of OPCs cryopreserved with the cryopreservation medium containing D(+)-trehalose dihydrate, which was comparable to that of non-frozen cells.
The study concluded that hNSC-induced OPCs collected by digestion and cultured adherently can be cryopreserved with the OPS complete medium containing 70 mL/L DMSO + 300 mL/L FBS + 0.2 mol/mL D(+)-trehalose dihydrate for a long time by slow cooling and rapid rewarming in the freezing box, with an average recovery rate of up to 75%. The recovery after cryopreservation did not affect OPC-specific marker expression and proliferation capacity.
D(+)-trehalose dihydrate is a non-toxic cell-membrane-impermeable cryoprotectant from many sources, which has been proven to have a good cryopreservation effect for various cell types such as hPBSCs and hESCs. In this study, the cryopreservation medium supplemented with low concentration of D(+)-trehalose dihydrate achieved the highest recovery rate in all experimental groups, and the results of proliferation and marker expression after culture indicated that the cryopreservation medium added with D(+)-trehalose dihydrate were also suitable for cryopreservation of human OPCs and could improve the recovery rate of the cells.